

2024 , Vol. 20 >Issue 03: 346 - 352
DOI: https://doi.org/10.3877/cma.j.issn.1673-5250.2024.03.014
重组人生长激素治疗特发性矮小症患儿的疗效
收稿日期: 2023-12-08
修回日期: 2024-05-09
网络出版日期: 2024-08-19
版权
Efficacy of recombinant human growth hormone therapy in children with idiopathic short stature
Received date: 2023-12-08
Revised date: 2024-05-09
Online published: 2024-08-19
Copyright
探讨重组人生长激素(rhGH)治疗特发性矮小症(ISS)患儿的疗效。
选择2020年10月至2022年12月于潍坊医学院附属医院儿科门诊就诊的84例ISS患儿为研究对象。首先根据是否采取rhGH治疗,将其分为治疗组(n=45,注射rhGH者)和对照组(n=39,未注射rhGH者)。其次,再根据患儿年龄,将治疗组分为治疗亚组1(n=21,年龄<8岁者),治疗亚组2(n=24,年龄≥8岁者),进一步将对照组分为对照亚组1(n=19,年龄<8岁者)和对照亚组2(n=20,年龄≥8岁者)。观察治疗组和对照组患儿的性别、年龄、身高、生长速率(GV)、骨龄、胰岛素样生长因子(IGF)-1、25羟维生素D[25(OH)D]及空腹血糖(FBG)水平。本研究遵循的程序符合潍坊医学院附属医院伦理委员会批准(审批文号:wyf-2022-ky-217),所有患儿及家属对本研究充分知晓,并签署知情同意书。
①治疗前,治疗组和对照组患儿年龄、身高、GV、骨龄及IGF-1、25(OH)D及FBG水平比较,差异均无统计学意义(P>0.05)。②治疗2年后,治疗组和对照组患儿身高、GV、IGF-1和25(OH)D水平比较,差异均有统计学意义(t=3.80、17.27、6.34、2.34,P=0.006、0.008、<0.001、<0.001),而骨龄、FBG比较,差异均无统计学意义(P>0.05)。③治疗2年后,治疗亚组1、2患儿的身高和年GV分别优于对照1、2组,并且差异均有统计学意义(t=5.00、64.35,P=0.007、0.009)。此外,治疗亚组1患儿的年GV大于治疗亚组2,并且差异有统计学意义(t=35.60,P=0.006);而对照亚组1、2患儿的年GV比较,差异无统计学意义(P>0.05)。④治疗1年后,4个亚组患儿GV比较,治疗亚组2于治疗后7~12个月的GV明显高于0~6个月,组内比较,差异有统计学意义(t=0.37,P=0.049)。⑤研究组与对照组患儿治疗过程中出现关节疼痛、肝肾功能异常、甲状腺功能异常、FBG升高等总发生率比较,差异均无统计学意义(P>0.05)。
rhGH治疗ISS患儿,可显著提高其身高和GV,尤其在<8岁儿童中效果更佳,并且治疗过程中未发现严重不良反应。
李倩 , 刘倩 , 朱海玲 , 倪娟 , 任宝芹 , 刘长云 . 重组人生长激素治疗特发性矮小症患儿的疗效[J]. 中华妇幼临床医学杂志(电子版), 2024 , 20(03) : 346 -352 . DOI: 10.3877/cma.j.issn.1673-5250.2024.03.014
To explore the efficacy of recombinant human growth hormone (rhGH) in the treatment of idiopathic short stature (ISS).
A total of 84 children with ISS diagnosed from October 2020 to December 2022 at the pediatric outpatient department of Weifang Medical College Affiliated Hospital were selected into this study. They were divided into treatment group (n=45, those who received rhGH injections) and control group (n=39, those who did not receive rhGH injections). Further stratification was done based on the children′s age into treatment group 1 (n=21, children under 8 years old), treatment group 2 (n=24, children aged 8 years or older), control group 1 (n=19, children under 8 years old), and control group 2 (n=20, children aged 8 years or older). The gender, age, height, growth velocity (GV), bone age, insulin-like growth factor (IGF)-1, 25-hydroxyvitamin D [25(OH)D], and blood glucose levels of the children were observed. The procedures of this study were in accordance with the approval of the Ethics Committee of Weifang Medical College Affiliated Hospital (Approval No.wyf-2022-ky-217), and informed consents were obtained form all children and their families.
① There were no statistically significant differences in age, height, GV, bone age, IGF-1, 25(OH)D, and blood glucose levels between the treatment and control groups before treatment (P>0.05). ② After 2 years of treatment, there were statistically significant differences in height, GV, IGF-1, and 25(OH)D levels between treatment and control groups (t=3.80, 17.27, 6.34, 2.34; P=0.006, 0.008, <0.001, <0.001), while there were no significant differences in bone age and blood glucose levels (P>0.05). ③ After 2 years of treatment, the height and annual GV of children in treatment groups 1 and 2 were significantly better than those in control groups 1 and 2, respectively, and the differences were statistically significant (t=5.00, 64.35; P=0.007, 0.009). Moreover, the annual GV of children in treatment group 1 was greater than that in treatment group 2, and the difference was statistically significant (t=35.60, P=0.006). However, there was no significant difference in the annual GV of children in control groups 1 and 2 (P>0.05). ④ When comparing the GV of children in each subgroup after 1 year of treatment, the GV of treatment group 2 during months 7 to 12 was significantly higher than that during the first 6 months, and the difference was statistically significant within the group (t=0.37, P=0.049). ⑤ There were no statistically significant differences in the incidence of joint pain, abnormal liver and kidney function, thyroid dysfunction, and increased blood sugar during the treatment process between the two groups (P>0.05).
Treatment with rhGH can significantly improve the height and GV of children with ISS, especially in those under 8 years old, and no serious adverse reactions were found during the treatment.
表1 治疗前,治疗组和对照组ISS患儿一般临床资料比较 |
| 组别 | 例数 | 性别[例数(%)] | 年龄(岁,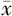 ±s) ±s) | 身高(cm,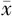 ±s) ±s) | GV(cm/年,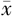 ±s) ±s) | 骨龄(岁,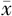 ±s) ±s) | IGF-1(ng/mL,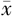 ±s) ±s) | 25(OH)D(ng/mL,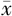 ±s) ±s) | |
|---|---|---|---|---|---|---|---|---|---|
| 男 | 女 | ||||||||
| 治疗组 | 45 | 22(48.9) | 23(51.1) | 8.1±2.8 | 116.3±14.7 | 4.2±0.4 | 7.1±2.0 | 160.4±19.3 | 17.8±1.3 |
| 对照组 | 39 | 19(48.7) | 20(51.3) | 8.0±2.9 | 114.1±14.2 | 4.2±0.4 | 7.0±2.8 | 158.4±17.8 | 17.9±1.0 |
| 统计量 | χ2<0.001 | t=0.14 | t=0.68 | t=0.51 | t=0.27 | t=0.43 | t=0.36 | ||
| P值 | >0.999 | 0.892 | 0.497 | 0.610 | 0.786 | 0.653 | 0.719 | ||
注:治疗组为接受常规治疗+rhGH治疗措施的ISS患儿,对照组为仅采取常规治疗措施的ISS患儿。ISS为特发性矮小症,GV为生长速率,IGF-1为胰岛素样生长因子1,25(OH)D为25羟维生素D,rhGH为重组人生长激素 |
表2 治疗前及治疗2年后,治疗组和对照ISS患儿GV和骨龄比较( |
| 组别 | 例数 | 身高(cm) | GV (cm/年) | 骨龄(岁) | IGF-1(μg/L) | FBG(mmol/L) | 25(OH)D (ng/mL) |
|---|---|---|---|---|---|---|---|
| 治疗组 | 45 | 134.1±12.2 | 8.9±1.7 | 9.8±2.8 | 210.7±1.2 | 4.3±0.5 | 29.9±2.1 |
| 对照组 | 39 | 123.0±14.5 | 4.4±0.3 | 8.8±2.8 | 170.2±1.2 | 4.4±0.5 | 23.5±2.4 |
| t值 | 3.80 | 17.27 | 1.49 | 6.34 | 0.25 | 2.34 | |
| P值 | 0.006 | 0.008 | 0.139 | <0.001 | 0.762 | <0.001 |
注:治疗组为采取常规治疗+rhGH治疗措施的ISS患儿,对照组为仅采取常规治疗措施的ISS患儿。ISS为特发性矮小症,GV为生长速率,IGF-1为胰岛素样生长因子1,FBG为空腹血糖,25(OH)D为25羟维生素D,rhGH为重组人生长激素 |
表3 治疗前和治疗2年后,不同年龄段4个亚组ISS患儿身高、GV、骨龄比较( |
| 项目 | <8岁患儿 | ≥8岁患儿 | ||||||
|---|---|---|---|---|---|---|---|---|
| 治疗亚组1(n=21) | 对照亚组1(n=19) | t值 | P值 | 治疗亚组1(n=24) | 对照亚组1(n=20) | t值 | P值 | |
| 身高(cm) | ||||||||
| 治疗前 | 103.1±9.3 | 101.4±8.3 | 0.59 | 0.555 | 127.9±6.7 | 126.2±5.4 | 0.87 | 0.392 |
| 治疗2年后 | 124.5±9.6 | 109.9±8.2 | 5.00 | 0.007 | 142.5±6.7 | 135.3±5.7 | 3.73 | 0.008 |
| GV (cm/年) | ||||||||
| 治疗前 | 3.9±0.3 | 3.9±0.2 | 0.89 | 0.381 | 4.5±0.4 | 4.5±0.2 | 0.01 | 0.993 |
| 治疗2年后 | 10.7±0.4 | 4.3±0.2 | 64.35 | 0.009 | 7.4±0.2 | 4.6±0.3 | 39.29 | 0.006 |
| 骨龄(岁) | ||||||||
| 治疗前 | 4.7±1.3 | 4.5±1.3 | 0.39 | 0.696 | 9.3±1.6 | 9.3±1.5 | 0.04 | 0.965 |
| 治疗2年后 | 7.0±1.5 | 6.8±1.3 | 1.97 | 0.157 | 11.9±1.6 | 11.2±1.6 | 1.56 | 0.127 |
注:治疗亚组1和对照亚组1为年龄<8岁,分别采取常规治疗+rhGH治疗措施与仅采取常规治疗措施的ISS患儿,治疗亚组2和对照亚组2为年龄≥8岁,分别采取常规治疗+rhGH治疗措施与仅采取常规治疗措施的ISS患儿。GV:治疗亚组1 vs治疗亚组2,t=35.60,P=0.006;对照亚组1 vs对照亚组2,t=0.12,P=0.908。ISS为特发性矮小症,GV为生长速率,rhGH为重组人生长激素 |
表4 治疗0~6个月与7~12个月时,4个亚组患儿GV组内比较(cm/年, |
| 组别 | 例数 | GV | t值 | P值 | |
|---|---|---|---|---|---|
| 治疗0~6个月 | 治疗7~12个月 | ||||
| 治疗亚组1 | 21 | 5.4±0.4 | 5.4±0.3 | 0.28 | 0.778 |
| 治疗亚组2 | 24 | 3.5±0.2 | 4.2±0.2 | 0.37 | 0.049 |
| 对照亚组1 | 19 | 2.4±0.1 | 2.3±0.2 | 2.29 | 0.058 |
| 对照亚组2 | 20 | 2.3±0.2 | 2.3±0.2 | 1.15 | 0.258 |
注:治疗亚组1和对照亚组1为年龄<8岁,分别采取常规治疗+rhGH治疗措施与仅采取常规治疗措施的ISS患儿,治疗亚组2和对照亚组2为年龄≥8岁,分别采取常规治疗+rhGH治疗措施与仅采取常规治疗措施的ISS患儿。ISS为特发性矮小症,GV为生长速率,rhGH为重组人生长激素 |
表5 治疗组和对照组ISS患儿治疗过程中不良反应总发生率比较[例数(%)] |
| 组别 | 例数 | 关节疼痛 | 肝肾功能异常 | 甲状腺功能异常 | FBG升高 | 总发生率 |
|---|---|---|---|---|---|---|
| 治疗组 | 45 | 2(4.4) | 1(2.2) | 1(2.2) | 1(2.2) | 5(11.1) |
| 对照组 | 39 | 2(5.1) | 1(2.6) | 0(0) | 1(2.6) | 4(10.2) |
| χ2值 | — | — | — | — | 0.02 | |
| P值 | — | — | — | 0.089 |
注:治疗组为采取常规治疗+rhGH治疗措施的ISS患儿,对照组为仅采取常规治疗措施的ISS患儿。"-"表示未进行统计学比较。ISS为特发性矮小症,rhGH为重组人生长激素 |
利益冲突 所有作者声明无利益冲突
| [1] |
|
| [2] |
|
| [3] |
学龄前儿童(3~6岁)运动指南编制工作组,关宏岩,赵星,等. 学龄前儿童(3~6岁)运动指南[J].中国儿童保健杂志,2020, 28(6): 714-720. DOI: 10.11852/zgetbjzz2020-0982.
|
| [4] |
王瑞青,孔宪菲,张华,等. 世界卫生组织身体活动和久坐行为指南[J].中国卒中杂志,2021, 16(4): 390-397. DOI: 10.3969/j.issn.1673-5765.2021.04.012.
|
| [5] |
|
| [6] |
曾佩佩,黄丽萍,李红辉,等. 特发性矮小症患儿心理行为问题分析[J].医学信息,2020, 33(23): 119-121. DOI: 10.3969/j.issn.1006-1959.2020.23.035.
|
| [7] |
|
| [8] |
陈凌华,赵慧,张素珍,等. 矮小症儿童主观生活质量和社会适应能力的研究[J]. 中国儿童保健杂志,2016, 24(12): 1271-1273. DOI: 10.11852/zgetbjzz2016-24-12-11.
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
雷巧容,李自尊,宋芬芳,等. 生长激素释放肽基因多态性与青春期前特发矮小患儿重组人生长激素治疗疗效的关系研究[J].实用医院临床杂志,2022,19(6):143-147. DOI: 10.3969/j.issn.1672-6170.2022.06.041.
|
| [13] |
王克成,唐海俊,沈莉. 不同病因矮小症患儿血清生长激素释放肽、胰岛素样生长因子-1水平变化及其临床意义[J]. 发育医学电子杂志,2024, 12(3): 179-186. DOI: 10.3969/j.issn.2095-5340.2024.03.004.
|
| [14] |
杨玉,黄慧,余珍,等. 江西省特发性矮小症与IGF-1R基因多态性相关性研究[J].中国儿童保健杂志,2015, 23(7): 710-712, 716. DOI: 10.11852/zgetbjzz2015-23-07-11.
|
| [15] |
|
| [16] |
陈志玫,刘静,孙利伟,等. 特发性矮小患儿矮小同源盒基因突变与血清胰岛素样生长因子-1水平关系的研究[J].中国妇幼保健,2020, 35(9): 1696-1699. DOI: 10.19829/j.zgfybj.issn.1001-4411.2020.09.040.
|
| [17] |
杨荣平,李娟,单良,等. 重组人生长激素治疗对小儿身材矮小血清IGF-1和Ghrelin水平及对体重指数影响 [J].临床和实验医学杂志,2021, 20(20): 2202-2205. DOI: 10.3969/j.issn.1671-4695.2021.20.021.
|
| [18] |
费丹宏. 重组人生长激素治疗特发性矮小症的疗效及对患儿生长发育的影响[J].中国妇幼保健,2021,36(6):1311-1314. DOI: 10.19829/j.zgfybj.issn.1001-4411.2021.06.031.
|
| [19] |
潘丹丹,刘建华,余瑶. 重组人生长激素治疗超大骨龄特发性矮小及偏矮儿童临床研究[J]. 中国现代药物应用,2021, 15(20): 178-180. DOI: 10.14164/j.cnki.cn11-5581/r.2021.20.070.
|
| [20] |
|
| [21] |
许可,宁刚. 基于人工智能的骨龄辅助评价系统对四川地区完全性生长激素缺乏症患儿骨龄研究 [J/OL]. 中华妇幼临床医学杂志(电子版), 2022, 18(4) : 449-459. DOI: 10.3877/cma.j.issn.1673-5250.2022.04.011.
|
| [22] |
|
| [23] |
邓茜,陈雨青,王娟娟,等. 特发性矮小症患儿治疗前后血清IGF-1、IGFBP-3、25(OH)D、皮质醇水平变化及其与体格发育和骨龄的相关性分析[J].现代生物医学进展,2022, 22(18): 3524-3527, 3595. DOI: 10.13241/j.cnki.pmb.2022.18.023.
|
| [24] |
陈丽娜,向承发. 重组人生长激素对身材矮小的小于胎龄儿身高的促增长作用 [J/OL]. 中华妇幼临床医学杂志(电子版), 2012, 8(5): 585-587. DOI: 10.3877/cma.j.issn.1673-5250.2012.05.007.
|
| [25] |
杨凡. "基因重组人生长激素儿科临床规范应用的建议"解读 [J/OL]. 中华妇幼临床医学杂志(电子版), 2014, 10(2): 141-144. DOI: 10.3877/cma.j.issn.1673-5250.2014.02.004.
|
/
| 〈 |
|
〉 |